Featured Disruptive Innovation Projects
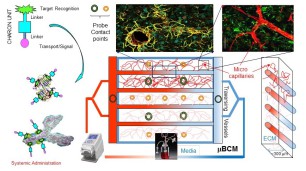
The Brain-Blood-Barrier (BBB) is a specialized protective system effectively blocking 98% of FDA-approved drug delivery into the brain. Our universal cargo ferrying platform design (CHARON) using non-competitive transporting mechanisms while leaving the BBB functionality intact, is aimed to change that (left).
The Artificially Perfused and Naturally Vascularized (APVN) platforms allow engineering multiple 3D cellular and tissue culture model types including neuronal co-cultures with mature tissue architecture.
The importance and economic impact of Type I and Type II (insulin dependent) Diabetes Mellitus represents an increasing human burden measured by hundreds of billions of $ and affecting over 350 million people worldwide. Our aim is to contribute to better disease management through development of new glucose responsive insulin forms that are capable of addressing the variety of shortcomings related to complex instability, administration, insulin overdose, glucose concentration sensing, and homeostatic insulin release; factors that are responsible for maintaining physiological circulating glucose concentration control at the molecular level.
The following video clip below illustrates our design approach to Glucose Responsive Insulin development.
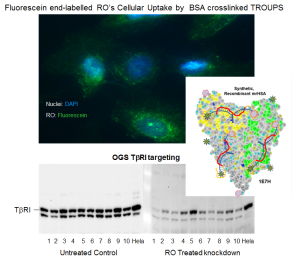
Therapeutic oligonucleotides carry a great potential to fundamentally simplify interventions if their design and delivery would be able to ensure that they are cell, organ, target molecule specific, without any significant side effect. The Regulatory Oligonucleotides (RO can be any/multiple types such as aptamers, RNAi, ASO, shRNA, siRNA, anti-piRNA, anti-lncRNA, splicing regulators, ribozyme, etc.). Enter our Regulatory Oligonucleotide Platform System (TROUPS) facilitating RO Activated Release (ROAR). Our design comprised of multiple components, including a serum protein conjugated holding platform that carries the RO’s regulatory core sequence (by Watson-Crick base pairing) which is then flanked by cell and tissue specific targeting regions to be activated on site and inducing ROs release from the holding/carrier platform.
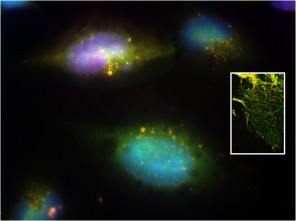
In vivo, Matrix Metalloproteinases (MMPs) in conjunction with their natural tissue inhibitors (TIMPS), play a fundamental role in maintaining extracellular matrix (ECM) architecture, remodeling, and global tissue homeostasis. Together, MMPs and TIMPs are key players in modulating tissue health and pathology and relevant microenvironmental changes (as for example in cellular invasion). Consequently, there is an important distinction between general MMP/TIMP secretion processes and directional cellular trafficking/transport in combination with local matrix deposition and/or activation. Cellular expression of MMP (red) and their directional trafficking (inset) are shown.
The following video clip below is a Total Internal Reflection Fluorescence Microscope (TIRFM) imaging of MMP1-dsRed cellular delivery to the cell-matrix interface (collagen-fibronectin) green viewed from below
Credits: We thank Zeiss and Kevin Brown to providing access to the demo TIRFM system at University of Miami, and the HIHG for hosting the demo co-managed by Justin Price HIHG-ISCI Zeiss LSM710 and George McNamara (UM). The experiment was performed by Nandor Garamszegi and imaged by George McNamara, currently is a Senior Research Scientist & Single Cells Imaging Analyst at University of Texas M.D. Anderson Cancer Center
Arbuscular mycorrhizal fungi (AMF) symbiosis has a great potential to significantly contribute sustainability and plant nutrition. This extensive, beneficial and mutualistic symbiosis involves over 75% of all known plant species. AMF represent one of the oldest fungal lineages, arguably being the most ecologically and agricultirally important terrestial symbiont affecting corp production on the earth. Our AMF collaboration is aimed to develop new approaches and widespread more effective AMF utilization for delivering real agricultural and economical sustainability.
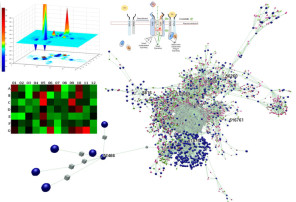
To better understand cell-matrix communication and its role in the homeostatic maintenance of organ-specific tissue architecture, we regularly perform focused gene expression studies coupled with relevant signal transduction analyses and interaction network modeling. This approach allows us to better understand, predict, and measure how the function of engineered matrix affects cellular gene expression profiles and to study how these matrices regulate tissue specific cellular phenotypes and biological functions. In exchange, we are in better position to manipulate developmental, pathological microenvironments (as for example from stem cell applications to tumorigenesis).
As an example of our ongoing tissue engineering efforts, we are researching novel methods to develop microscopic tendons (over 100-micron diameter and several centimeter lengths) from tendon fibroblast cell-sheets (shown). Our approach allows us to study the regulation of cellular interactions and phenotypes, attachment, and fiber formation, in different regions of the cellular matrix. Our results are directly transferable to macroscopic scales. Additional projects in specialized extracellular matrix development target multiple organ-specific architectures for bone, pancreas, neurological, and other model systems